In this project we’re interested in building up a picture of colonial people’s lives using their biology. But how are we actually doing that I hear you ask? In this series of blogs we’ll be looking at the techniques our group are using to get to the bottom of things. Or throw up more questions (as is more often the case).
First up we have chemical (or isotopic) techniques. My work!
As an archaeological chemist I know that as soon as I start talking about chemistry people’s eyes tend to glaze over. But your bone, tooth and hair chemistry can tell the archaeologist heaps of cool stuff about your life. When you’re alive your body is sourcing its building blocks for your tissues from the world around you. That’s from the food you eat, the water you drink, the air you breathe, and the ground you live on. That means by looking at your skeletal chemistry we can get an idea of what was going on in your life.
I’m working with lots of different isotope systems (an isotope is just an alternative form of an element that is a slightly different weight) to try and build up a good picture of people’s lives. Here’s a bit of a lowdown on each of them:
Strontium: Strontium is sourced from the geology in the place you live, or the place you grow your food at least. Strontium from local soils is in the plants and animals you eat, and it becomes incorporated into your body too. Your teeth form when you’re a child, so the strontium in your teeth will reflect the geology where you grew up. If it’s different from the place where you’re buried we can tell you’re a migrant. In our case, strontium ratios that don’t match the south of the South Island tell us that a person is probably a first settler. Exciting! And sometimes they can help us to narrow down where the person might be from originally too…
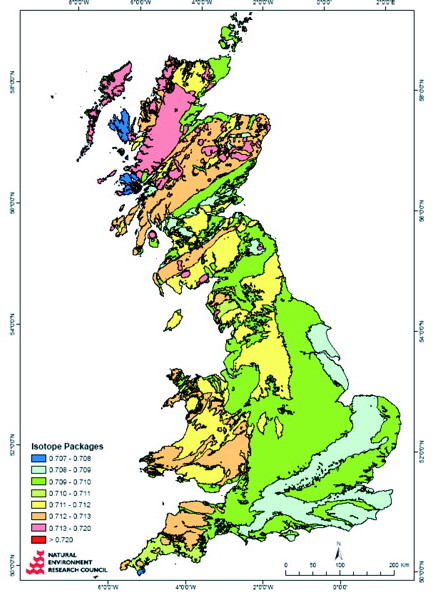
Lead: Lead is another isotope system that relates to where you’re from. Different rocks have different lead ratios. BUT lead is also released into the atmosphere as soon as you start mining rocks, or smelting metals. So if you’re from a place with lots of industry like, say, Industrial Revolution period Europe, you’ll have way more lead in your system than someone from non-industrialised New Zealand. So lead can help us to tell if a person is a first European settler too.

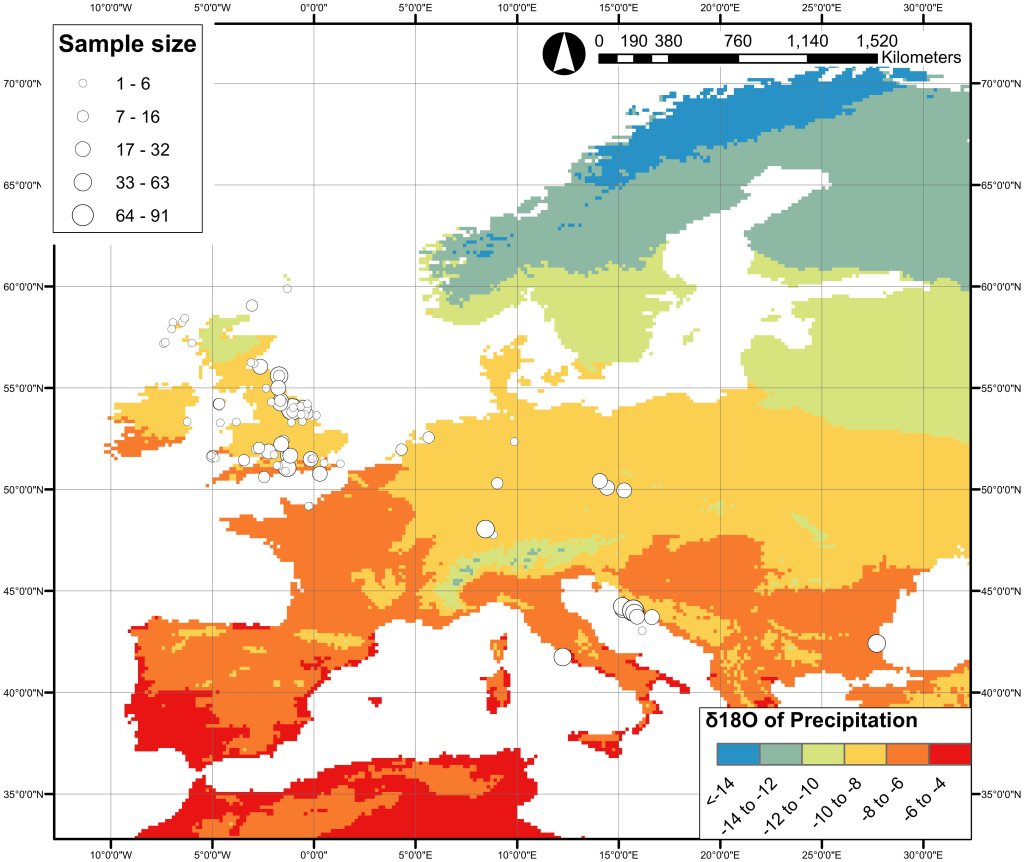
Oxygen: Water is full of oxygen (#chemistryfacts). So it’s no surprise that oxygen isotope ratios in your tissues mostly tell us about the water you were drinking during life, specifically what kind of environment it came from. If you’re from a rainy place your oxygen isotope ratios will be different to someone from a hot, dry place. If you’re from a big continent your isotope ratios will be different to someone who’s from a little island. To be honest, the oxygen isotope system is really complicated and even actual chemists don’t fully understand it. It’s always a bit difficult to interpret, but in our study it should help show up people who are from somewhere climatically different to Otago.
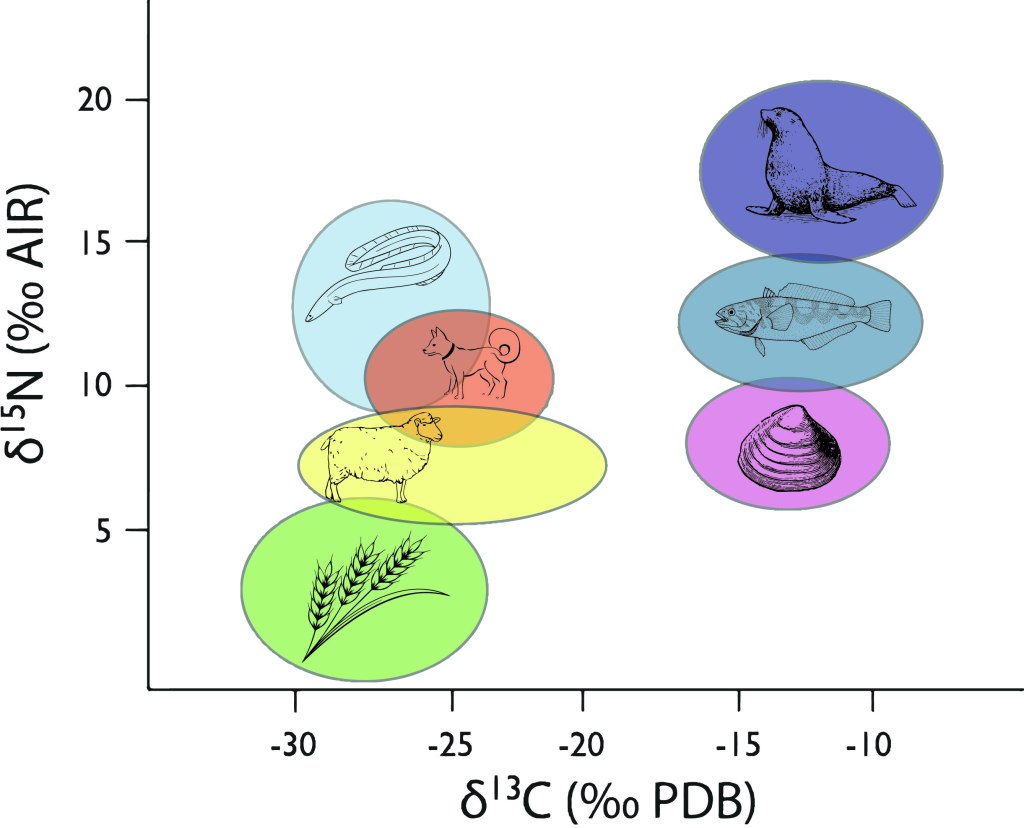
Carbon and Nitrogen: Carbon and nitrogen go together like lamb and mint sauce. Together they can tell us about both the plants and the animals that people were eating. Carbon isotopes mostly come from the plants you eat, and different types of plants have different carbon isotope ratios. Nitrogen mostly tells us about how much meat is in your diet, the higher your nitrogen isotope ratio, the more meat you’re eating. Oh, and if you’re eating seafood both your carbon and nitrogen isotope ratios change in known ways. So they tell us if you’re using the land, the sea, or a mixture of both to keep yourself fed.
So, lots of chemistry, lots of results to come, stay tuned!
Charlotte King
References:
Evans, J., Montgomery, J., Wildman, G., & Boulton, N. (2010). Spatial variations in biosphere 87 Sr/ 86 Sr in Britain. Journal of the Geological Society, 167, 1–4.
Lightfoot, E., & O’Connell, T. C. (2016). On the Use of Biomineral Oxygen Isotope Data to Identify Human Migrants in the Archaeological Record: Intra-Sample Variation, Statistical Methods and Geographical Considerations. PloS One, 11(4), e0153850.
5 thoughts on “It’s all about chemistry!”